Cell Signaling Technologyは、研究用途のみに使用可能な製品やサービスを提供しており、
これらは診断または治療には使用できません。
キメラ抗原受容体 (CAR) 細胞療法は、B細胞性白血病やリンパ腫などの血液がんに対して驚異的な有効性を示す、急速に発展しているがん治療法です。 しかし、この治療法の固形がんへの応用には依然として課題があります。2025年4月現在、固形がんに対するCAR-T細胞療法やCAR-NK細胞療法は、米国で複数が臨床試験中ではあるものの、規制当局の承認を取得したものはまだありません1,2,3。
本ブログ記事では、固形がんの免疫療法におけるCARベースの細胞療法の活用に向けた最新の戦略を探索し、Whitlow/218リンカーを含む単鎖可変フラグメント (scFv) ベースのCAR改変細胞の空間的な解析を可能にする新たな抗体、Whitlow/218 Linker (F2G3S) Rabbit mAb #47414を紹介します。現在、この抗体は免疫組織化学染色 (IHC) アッセイで検証済みです。
有望な可能性:固形がんの免疫療法における課題の克服
固形がんは、生物学的背景が血液がんとは根本的に異なります。固形がんは、がん細胞が血液中を自由に循環する白血病やリンパ腫とは異なり、免疫細胞の浸潤を抑制し、その活性化を防止、さらに免疫細胞による攻撃からがん細胞を保護するために特別に組織された、非常に複雑な細胞環境を構築します。
また、血液がんではCD19やBCMAといった細胞タイプに特異的な表面抗原がCARベースの細胞療法の明確な標的となりますが、固形がんでは腫瘍抗原の発現が不均一であるため、各腫瘍タイプに対して複数の抗原を治療標的として戦略的に選択する必要があります。適切な標的抗原を特定することが難しいだけでなく、同じがんタイプの患者間でも腫瘍抗原の発現が異なるため、ある患者に対して有効な標的抗原パネルが、別の患者には適さない場合もあります。この課題をさらに複雑にしているのは、固形がんで発現する多くの標的抗原が正常組織でも発現しているという事実であり、これによりオンターゲット/オフ腫瘍毒性、すなわち患者に深刻な安全上のリスクが生じる可能性があります1,4。
CSTが提供するビデオやポスターを探索し、CAR改変細胞の特性をより簡単かつより迅速に特性解析する方法をご覧ください。
これらの問題などに対処するため、研究者は様々なアプローチを試みています。有望な戦略の1つに、AND論理ゲートに基づくCARシステムの活用があります。この手法では、異なる抗原を標的とする2種類のCARを発現するように改変されたT細胞は、その両方のCARがそれぞれの標的抗原と結合した場合にのみ活性化されます。この二重抗原認識により、特異性が向上し、健康な組織への損傷のリスクが低減します。
がん微小環境 (TME) に侵入したCAR-T細胞を保護する戦略も探索されています。この戦略では、CAR-T細胞がTGF-βなどの免疫抑制性サイトカインへの抵抗性を獲得できるように、CAR-T細胞に対してドミナントネガティブ受容体を発現させる遺伝子改変が行われています。
特異性を向上させて免疫抑制を克服する戦略を取り入れた、固形がんに対する有望なCAR細胞療法をいくつか紹介します。
- 拡散性中線膠芽腫 (DMG) に対するGD2を標的とするCAR細胞療法:小児患者における早期臨床試験で、腫瘍の縮小と神経機能の改善がみられます6。
- 消化器がんと肺がんに対するCEAを標的とするCAR細胞療法1
- 胃がんや膵がんに対するClaudin18.2特異的CAR細胞療法:ただし、現時点での臨床データは初期的なものです1。
固形がんにおけるCAR-T細胞の空間的な解析
研究者により、CARの改変戦略は改良され続けていますが、依然として「これらの細胞は、実際に腫瘍内の標的部位に到達しているのか?また、有効な攻撃を行えるだけの十分な期間、そこに存在し続けているのか?」という疑問が残されています。この疑問に答えるために、研究者は、がん微小環境内のCAR改変細胞の空間的配置を可視化し定量できるツールを必要としています。
組織内のCAR-T改変細胞を検出する方法をいくつか紹介します。
抗イディオタイプ抗体
抗イディオタイプ抗体は、長年用いられているCAR検出のゴールドスタンダードです。しかし、これらの抗体は通常、特定のCARコンストラクト用にカスタム作製されており、その開発には時間とコストがかかります。さらに、CAR構造の改変 (例えば、結合特性の最適化など) により、抗イディオタイプ抗体が認識するエピトープが変化し、検出できなくなる可能性があります。
RNA in situハイブリダイゼーション (RNA ISH)
もう1つのよく用いられる手法に、RNA in situハイブリダイゼーション (RNA ISH) があります。この手法では、組織内のCARのmRNA転写産物を検出します。この技術を用いることにより、研究者は、貴重な空間的情報を取得して腫瘍内におけるCAR-T細胞の局在を可視化できます。ただし、これには重大な欠点があります。この技術では、RNAの存在が確認できるだけであり、それが実際に機能的なCARタンパク質に翻訳されているかどうかを判断することはできません。TMEにおける複雑な翻訳後調節メカニズムにより、研究者は、目的の細胞が実際に存在し、腫瘍を活発に攻撃しているかどうかについて確信を持てずにいます。
抗CARリンカー抗体
抗CARリンカー抗体は、scFvベースのCAR改変細胞の存在をIHCアッセイで検出できる新たなタイプの試薬です。この試薬は、不均一なTMEにおける空間生物学を保持したままCAR改変細胞を検出できます。抗CARリンカー抗体は、抗イディオタイプ抗体とは異なり、多くのscFvベースのCARに存在する普遍的なペプチドリンカー (G4SとWhitlow/218) を標的とするため、リンカー構造以外に改変を加えたCARの検出も可能です。つまり、研究者は、異なる抗原を標的とするCARを設計および試験している場合でも、たった1つの試薬で複数のプログラムにおける検出を行うことができます。さらに、遺伝物質の存在量のみが得られるRNAベースの手法とは異なり、Whitlow/218リンカーラビット抗体はCARタンパク質を直接検出できるため、改変した細胞が細胞表面にCAR分子を積極的に発現しているかどうかを確認できます。
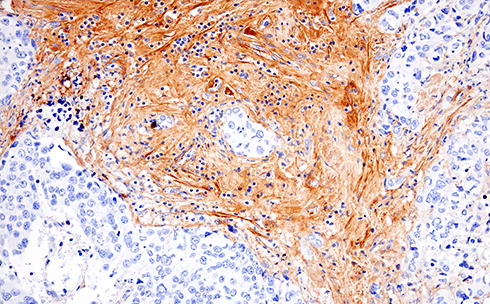
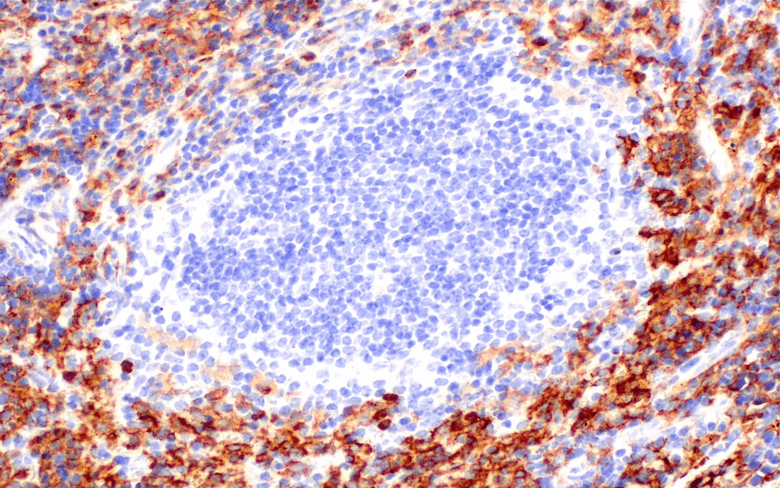
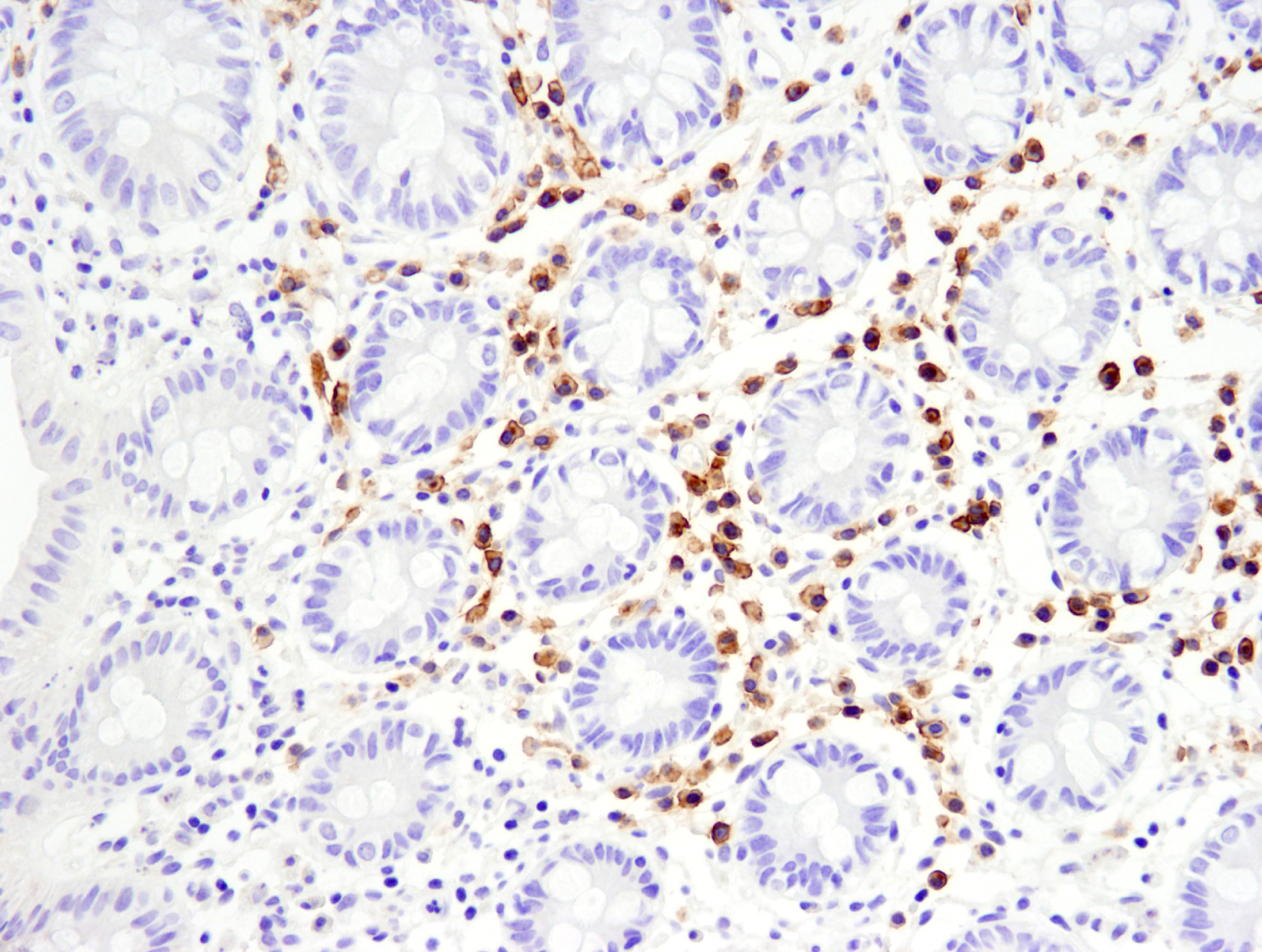
/Whitlow218%20Linker%20recombinant%20antibody_spatial%20analysis%20CAR.png?width=1013&height=342&name=Whitlow218%20Linker%20recombinant%20antibody_spatial%20analysis%20CAR.png) Raji B細胞リンパ腫の免疫不全マウス (NSG系統) におけるFFPE脾臓組織を、リコンビナントモノクローナル抗体Whitlow/218 Linker (F2G3S) Rabbit mAb #47414を用いてIHCで解析しました。プライマリーヒトCD19 CAR-T細胞で処理 (左) またはPBSコントロールで処理 (右) しています。
Raji B細胞リンパ腫の免疫不全マウス (NSG系統) におけるFFPE脾臓組織を、リコンビナントモノクローナル抗体Whitlow/218 Linker (F2G3S) Rabbit mAb #47414を用いてIHCで解析しました。プライマリーヒトCD19 CAR-T細胞で処理 (左) またはPBSコントロールで処理 (右) しています。
Whitlow/218 Linker (F2G3S) ラビット抗体は、ホルマリン固定パラフィン包埋組織 (FFPE) において、特異性や感度が高く、再現性のある結果の取得を可能にするため、研究者は、改変したT細胞が腫瘍内に実際に浸潤し、TMEに存在しているかどうかを正確に評価できます。
組織内のG4Sリンカーを含むCARを検出する方法は?CSTの科学者は、IHCでの使用を目的とする抗G4Sリンカー抗体の検証に積極的に取り組んでいます。最新情報を定期的に確認するか、進捗を弊社にお問い合わせください。
固形がん免疫療法の今後の展望
固形がんのCAR療法は、有望な可能性を示していますが、克服すべき課題があります。論理ゲート制御システムの活用や免疫抑制メカニズムへの抵抗性の設計、新たな標的の同定により、研究者は新たな成功への道を切り開くことができます。Whitlow/218リンカー抗体などのツールは、CARの存在や局在、持続性の迅速な解析を可能にします。これにより研究者は、新たな治療戦略を素早く開発できます。
|
|
CST抗体を用いたTMEの解析CSTは、PD1、CD25、TIM3、CD4、LAG3、CD8など、重要な表現型バイオマーカーと機能的バイオマーカーの抗体を用いたTMEの解析に最適な、IHCで検証された未標識および標識抗体の幅広い製品ラインナップを提供しています。 SignalStar®multiplex IHC技術は、1つのパネルで最大8種類のバイオマーカーを可視化できます。 CSTが提供するCAR改変細胞の特性解析ソリューションをご覧ください。 |
|
根本的な治療法としての確立には至ってはいませんが、空間生物学と適応型細胞工学の融合は、CAR改変細胞を用いた固形がんの免疫療法を大きく変える可能性を秘めています。
その他のリソース
- CSTが提供する抗CARリンカー抗体を、様々な研究者がどのように研究に用いているかについては、ブログ:研究の総括:論文で使用されているCSTの抗CARリンカー抗体をご覧ください。
- CAR遺伝子改変細胞の特性評価ソリューションに関するダウンロード可能なリソース (T細胞活性化アッセイや標識抗体および未標識抗体を用いる免疫表現型解析、機能性抗体など) については、こちらのパンフレットをご覧ください。
参考文献
- Khan SH, Choi Y, Veena M, Lee JK, Shin DS. Advances in CAR T cell therapy: antigen selection, modifications, and current trials for solid tumors. Front Immunol. 2025;15:1489827. Published 2025 Jan 6. doi:10.3389/fimmu.2024.1489827
- Yeku O, Li X, Brentjens RJ. Adoptive T-Cell Therapy for Solid Tumors. Am Soc Clin Oncol Educ Book. 2017;37:193-204. doi:10.1200/EDBK_180328
- Peng L, Sferruzza G, Yang L, Zhou L, Chen S. CAR-T and CAR-NK as cellular cancer immunotherapy for solid tumors. Cell Mol Immunol. 2024;21(10):1089-1108. doi:10.1038/s41423-024-01207-0
- Guzman G, Reed MR, Bielamowicz K, Koss B, Rodriguez A. CAR-T Therapies in Solid Tumors: Opportunities and Challenges. Curr Oncol Rep. 2023;25(5):479-489. doi:10.1007/s11912-023-01380-x
- Punekar SR, Kirtane K, Stein MN, et al. EVEREST-1: A seamless phase 1/2 study of A2B530, a carcinoembryonic antigen (CEA) logic-gated Tmod CAR T-cell therapy, in patients with solid tumors associated with CEA expression also exhibiting human leukocyte antigen (HLA)-A*02 loss of heterozygosity (LOH). J Clin Oncol. 2024;42(16_suppl):TPS2698
- Monje M, Mahdi J, Majzner R, et al. Intravenous and intracranial GD2-CAR T cells for H3K27M+ diffuse midline gliomas [published correction appears in Nature. 2024 Dec;636(8043):E6. doi: 10.1038/s41586-024-08452-3.]. Nature. 2025;637(8046):708-715. doi:10.1038/s41586-024-08171-9


/Tools%20to%20Evaluate%20CAR-T%20Cell%20Signaling%20%26%20Activation_small.png?width=200&height=113&name=Tools%20to%20Evaluate%20CAR-T%20Cell%20Signaling%20%26%20Activation_small.png)
/SignalStar%20Multiplex%20IHC%20analysis%20Option%202.png?width=215&height=345&name=SignalStar%20Multiplex%20IHC%20analysis%20Option%202.png)